Introduction to Chemistry
Chemistry
- It describes how substances interact with one another. It is the fundamental science behind biology, pharmacology and all of the other life sciences.
- Indeed, the importance of chemistry itself cannot be understated. So it is a fundamental science into which medicinal chemistry is rolled.
- Without a knowledge of chemistry, you may learn what happens in these topics but you will not be able to fully understand why things happen.
The Atom
- As time goes by, the scientific knowledge of mankind has increased and matter has been found to be made up of smaller and smaller fundamental particles.
- Physicists are still investigating the structure of the atom and discovering new and smaller particles.
- Chemistry is about the movements of electrons. Nuclear physics is about how nuclear particles interact with each other.
- A knowledge of the latest developments in sub-atomic physics is not necessary to account for most chemical behavior.
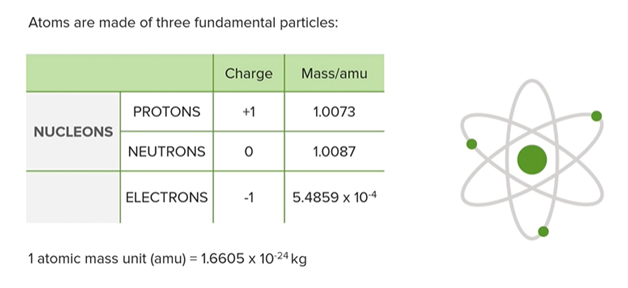
- The proton and the neutron are both nucleons which means that they are subatomic particles which reside within the nucleus of an atom.
- Chemistry is about electrons and not necessarily about nucleus.
- Most of the atom is empty space, the protons and the neutrons are clustered together in the center to form the nucleus
- The electrons seemingly form a cloud around the central nucleus. These electrons which engage with each other to form matter.
The Analogy
- The analogy which is often used for the structure of an atom is that the nucleus is the ball on the center spot and the electrons are specks of dust blowing around the stands.

- In atoms there are equal numbers of protons and electrons giving an overall charge of 0.
- Only certain combinations of fundamental practices can form stable atoms
- If an atom forms with an unstable composition in the nucleus, then it is liable to undergo some form of disintegration involving the loss of one or more fundamental practices which is known as RADIOACTIVE DECAY.
Atoms and Elements are defined by three numbers relating to the composition of the nucleus:
- Z– the atomic (or element) number
This is the number of protons in the nucleus and defines which element the atom is of.
- N– the neutron number
This is the number of neutrons
- A– the mass number
This is the number of protons added to the number of neutrons (Z+N)

- The atomic mass number is always larger than the atomic number.

- Sometimes, with periodic table, the Z is often omitted because the chemical symbol of a particular element automatically defined in the periodic table further defines the number of protons and electrons it must possess.
Periodic Table
- The periodic table lists all of the elements, both natural and man-made in order of increasing Z in rows moving left to right.

- As you can see in the table, you can see different numbers above and beneath the chemical symbols. The number above is the atomic number which correlates to the number of protons and the number of electrons that the element posseses in its atomic form.
- A lot of elements in nature don’t exist as atoms. They actually exists either as ions or they exist as molecules.

Electrons
- When the structure of the atom was established, the nucleus was found to be a solid mass of protons and neutrons with a positive charge and the negative electrons were found to surround it
How many electrons, protons, and neutrons are present in each isotope?

If positive charges attract negative charge, why don’t the electrons crash into the nucleus?
- The amount of energy which an electron has determines how far it is from the nucleus.
- That electrons can only be at certain, specific distances from the nucleus

Shells
- The energy levels in atom are called shells and these are numbered 1,2,3,4 from the lowest energy up. This number is known as the principal quantum number, n
. As the shells become larger they can hold more electrons.

Increasing Energy
- From a quantum perspective, each shell can carry a specific number of electrons.

- In order to move electron to a higher energy shell, a specific amount of energy is required- this amount of energy is called as a quantum. Electrons jumping down to a lower energy shell will emit a quantum of energy (light)

Quantum Mechanics
Ideas:
- Energy only comes in these fixed amounts or quanta
- Everything can be thought of as a particle or wave- the wave-like properties of a large object (people, items in the world around us) are insignificantly small.
- Related to this is the Uncertainty Principle and wave equations for electrons and other particles.
These theories mean when applied to the electrons in atoms, is that instead of being able to predict where they are or saying that they orbit the nucleus we can only say where there is the greatest probability of finding them.
Orbitals
- Shells can be further divided into orbitals, each of which can contain two electrons.
- Orbitals are characterized by the shape produced when the region surrounding the nucleus is plotted in which there is a 95% chance of finding the electrons.


p orbitals
- A p orbital has two regions where the electrons may be found either side of the nucleus.



d Orbitals
- (l = 2)
- Have more complex set of shapes and they have the orbital quantum
- Have two nodal planes and come in sets of five.
- ml can either be -2, -1, 0, 1, or 2
- lowest energy shell containing d orbitals is n=3
Degeneracy
- orbitals with same energy > 2
- These are orbitals with the same energy. They are superimposable onto each other
Quantum Numbers

- While building up a picture of shells and orbitals in atoms various quantum numbers were used to define them.
The 3 rules which are often used:
- The Pauli exclusion principle
- The Aufbau principle
- Hunds rule
The Pauli Exclusion Principle
- In any atom, no two electrons may have a set of quantum numbers with the same values.
- To obey the exclusion principle, we need to take into account a fourth quantum number which is to do with a property of the electron, not the orbital, called spin.
- Electrons can spin in one of the two directions represented by the magnetic spin quantum number
which can either be + ½ or – ½

- Electrons with the same M
are said to have parallel spin whilst those with different
values have anti-parallel spins. M
Aufbau Principle
- The Aufbau principle states that orbitals of the lowest energy are filled first.

Hund’s Rule
- It states that when filling a degenerate set or orbitals, electrons will not form pairs until all of the orbitals have a single electron in them.
- Elelctron in the singly filled orbitals will have parallel spins.

Valence Electrons
- Only the electrons in the outermost shell which participate during reactions are important for explaining an atom’s chemistry

- The remaining electrons in the core relate to the elements in the final group of the periodic table.
Bonding
- In nature systems, always seek to find their lowest energy. Most stable configuration.
- The most atomic configurations are those where the valence electron shell is completely filled.
Formation of Ions
- Atoms of other elements would like to gain stable configuration and attempt to do so by swapping or sharing electrons.
- For the first 18 elements a filled valence shell usually consists of 8 electrons (except H with 2), elements with higher atomic numbers require 18.
- Elements in groups 1, 2, 3 will tend to lose electrons whilst those in group 17 will tend to gain electrons.


Covalent Bonds
- Elements in groups towards the middle of the p-block of the periodic table do not tend to form ions
- These elements normally share, rather than exchange, electrons to achieve their full other shell.
Example:

- Due to their electron requirements, H will always form 1covalent bond, it is said to have a valency of 1
- C will always form 4 covalent bonds and it has a valency of 4.
Valence Bond Theory
- Electrons are found in orbitals, what happens to orbitals when electrons are shared during covalent bondings?

- The new orbital is a sigma orbital and it contains the nuclei of both atoms

- The better the overlap of the orbitals the stronger the bond.
- Both of the orbitals must have the same phase.
Other Sigma Bonds

- Both atoms can gain the extra electron that they need to get a fuller outer shell by sharing
Non Bonding

Electronegativity
- Atoms have varying affinities for electrons in chemical bonds. The affinity is described as electronegativity and it is measured on the Pauling scale.

- Large atoms have low affinity because the electrons are a long way from the positive nucleus, the core electrons shield the outer electrons from the nuclear charge
- In the periodic table, the affinity for bonding electrons increases because nuclear charge increases without a significant change in the distance of the valence electrons from the nucleus
Another Sigma Bond

Hybridization- Chemical Bonding
- The creation of s bonds explains the bonding in diatomic (2 atom) molecules but the situation is more complex with molecules containing more atoms.
- C has 2 p orbitals with single electrons able to form sigma bonds
- These orbitals will have 90 degrees between them
- In reality, the valence orbitals of C are mixed up or hybridized to give 4 orbitals suitable to form 4 sigma bonds


- Atoms other than C can also hybridize
- Knowledge of hybridization of orbitals gives information about the shapes of molecules.
Pi Bonds- Chemical Bonding
- Due to the shapes of p orbitals they can overlap in another way.
- Geometry and phase of the bonding orbitals must be correct
- Bonds formed in this way are called pi bonds and are formed by orbitals perpendicular to the bond axis.
- It always almost occurs between atoms which are already sigma bonded
Double and Triple Bond- Chemical Bonding
The double bond
- The p orbitals overlap to give a pi bond and simultaneously the sp2 orbitals overlap to give a sigma bond
- The remaining sp2 orbitals form a sigma bonds with the H atoms
The triple bond
- The sp orbitals will make a sigma bonds either to C or H and the p orbitals will overlap to give 2, perpendicular pi bonds
- There are 3 bonds between C atoms- a triple bond

- Hybridization and bonding in molecules not only affects geometry but also size and bond energies.
Stereochemistry and Isomerism
- Take their name from their effect on plane polarized light
- They have optical activity
- Molecules or ions that exist as optical isomers are said to be chiral
- Isomers with the same order of attachment of atoms in their molecules, but a different orientation of their atoms or groups of atoms in space.
- Configurational isomers are stereoisomers that do not readily interconvert at room temperature and can be separated.
- Conformational isomers are stereoisomers produced by rotation about sigma bonds.
Chirality
- Mirror images of one another and non-superimposable
- From Greek word ceir (cheir) for hands
- Chiral objects which is often described as left or right-handed
Chirality and Enantiomers
- The main structural feature responsible for chirality is the presence of dissymmetry in a molecule
- A carbon atom carrying four different atoms or groups is an element of dissymmetry. Such carbon atom is called the chiral centre.
- Carbon atom carrying four different groups has no plane of symmetry and determines therefore a chiral molecule
- DNA is chiral
- Small and taste receptors in our body are chiral
Cahn-Ingold-Prelog Rules
Naming:
- Rank atoms directly attached to the chiral center according to the atomic number.
- If there is a similarity at any substituent, look at the second, third, etc. until a difference is found.
- Multiple bonds as multiples substituents at that same atom.
- Rotate the lowest priority group to the back.
Identifying Enantiomers and Synthesis of Chiral Compounds
Identifying enantiomers
- There are no chemical nor physical differences between two enantiomers except for the following:
- Enantiomers rotate plane polarised light in equal degrees and opposite directions (hence the name optical isomers)
- Enantiomers react differently with chiral compounds
Keep in mind:
- (+)-enantiomers; rotate plane-polarised light to the right
- (-)-enantiomers: rotate plane polarized light to the left
- A racemic mixture does not rotate the plane of polarized light.
Electronegativity
Affinity for negative charge
- Large atoms have low affinity because the electrons are a long way from the positive nucleus, the core electrons shield the outer electrons from the nuclear charge.
- Going across the periodic table the affinity bonding electrons increases because nuclear charge increases without a significant change in the distance of the valence electrons from the nucleus.
Bond Polarization
- The most electronegative atom is fluorine with a value of 4.0 (group 18 elements in the periodic table do not react)
- All other elements have smaller Pauling values
- In bonds between atoms of different elements, the bonding electrons will be shared unequally
Dipoles- Electronegativity
- Electronegativity can give reasons for why chemical reactions occur
- In multiple dipoles, electrons may not evenly distribute in chemical bonds
- There are two ways in which chemical groups can alter the distribution of electrons (i) Inductive (ii) Resonance (mesomeric)
- The inductive effects are similar to the electronegativity effects of single atoms
- Functional groups can be electron-donating or electron-withdrawing (high electronegativity)
Observable Polarity – Electronegativity
- The effects can be felt through bonds and through space.

Resonance: Mesomeric Effects
- Pairs of valence electrons can move within molecules or between molecules, breaking and forming chemical bonds
- The movement of electron pairs are represented by curly arrows
- Resonance structures only differ in their electron distribution- their atoms do not move
- A double-headed arrow is only used between resonance structures
Examples of Resonance
- For resonance to occur, there needs to be a group donating and a group accepting a pair of electrons

Induction and Resonance
- Examples of groups which donate electron pairs in resonance structures represented by +M
- Any group with negative charge or a Ione pare of electrons
- Sometimes both resonance and inductive effects are present in the same molecule
- Some functional groups can have one type of inductive effect and the opposite resonance effect.
